
Biotech is the rare industry where a single decision — a missed FDA meeting, a flawed CMC package, an unvalidated assay — can compress an entire company’s runway. That is why biotech consulting has quietly become one of the most strategic line items on a modern life sciences budget. Founders no longer hire consultants only when something breaks; they hire them to keep things from breaking in the first place.
This guide explains what biotech consulting covers in 2026, who buys it, and where the field is heading as AI, capital constraints, and tightening regulatory expectations reshape drug development.
What Is Biotech Consulting?
Biotech consulting is the practice of advising organizations that research, develop, and commercialize biotechnology products on how to improve their R&D, regulatory, manufacturing, and commercial outcomes. Per Wikipedia’s definition, the discipline emerged from general management consulting because biopharma’s mix of deep science and complex regulation rewards specialists over generalists.
In practice, a biotech consultant might help a Series A oncology startup design its first-in-human trial on Monday, write a CMC strategy for a gene therapy IND on Tuesday, and run commercial diligence for a venture fund on Wednesday. The work spans the full development lifecycle, but it shares a single goal: turn science into approved, reimbursed, and scalable products without burning capital on avoidable mistakes.
Who Hires Biotech Consultants?
Three buyer profiles dominate:
- Emerging biotechs (pre-clinical to Phase 2) that run lean and need senior expertise on demand rather than full-time hires.
- Established biopharma (Pfizer, Moderna, Merck, AstraZeneca) that bring in consultants for pipeline strategy, launch planning, and post-merger integration.
- Investors — venture capital, private equity, and corporate VCs — who commission technical and commercial diligence before writing checks.
Biotech firms tend to focus on early-stage R&D and run lean on the business side, which is why outside support for commercial activities is so common in the industry.
5 Core Services Biotech Consultants Provide
The deliverables vary by firm, but most engagements fall into five categories.
1. Regulatory Strategy and Submissions
This is the bread and butter. Consultants help companies plan and execute pre-IND meetings, IND/CTA submissions, BLA/NDA filings, FDA Type B and Type C interactions, and global regulatory pathways through EMA, MHRA, and PMDA. Many regulatory consultants are former FDA reviewers, and that institutional fluency often shaves months off review cycles. For a deeper look at how regulatory work integrates with the broader pharma value chain, see our guide to pharmaceutical consulting.
2. Clinical Development
Trial design, protocol writing, biomarker strategy, site selection, CRO oversight, and adaptive trial methodology. With the average new drug now costing more than $1 billion to develop according to the Congressional Budget Office, the cost of a poorly designed Phase 2 trial is enormous, and clinical consultants exist to prevent that.
3. CMC, Manufacturing, and Quality
Chemistry, Manufacturing, and Controls work covers process development, analytical method validation, tech transfer, GMP readiness, and sterility assurance. Cell and gene therapy programs in particular lean heavily on CMC consultants because the manufacturing science is still maturing and regulators are still defining expectations.
4. Commercial Strategy and Market Access
Forecasting, pricing, payer strategy, launch planning, and competitive positioning. This is the domain where firms like McKinsey, BCG, L.E.K., and ClearView Healthcare Partners have built large practices, often working alongside boutique specialists who bring deeper therapeutic-area expertise.
5. Transaction and Investor Support
Due diligence, valuation support, expert witness work, and fundraising materials. Diligence engagements have become more frequent as biotech’s share of U.S. startup investment has fallen to a 20-year low of about 8%, putting more weight on every individual deal that does close.
The Market Backdrop: Why Demand Is Rising
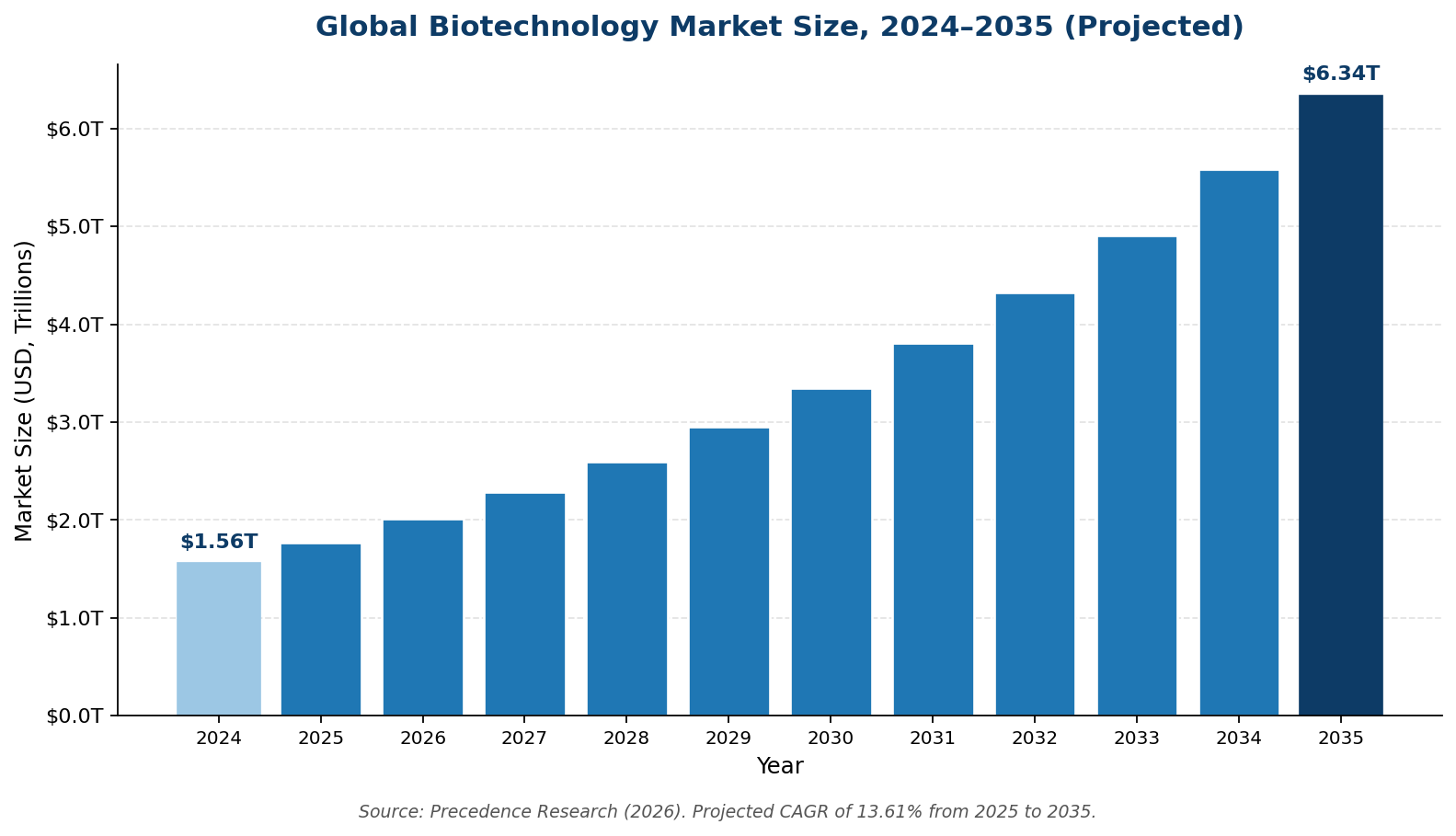 The biotechnology sector itself is the underlying driver of consulting demand. Multiple market research firms project the global biotech market will roughly triple over the next decade.
The biotechnology sector itself is the underlying driver of consulting demand. Multiple market research firms project the global biotech market will roughly triple over the next decade.
According to Precedence Research, the global biotechnology market was estimated at $1.77 trillion in 2025 and is projected to reach $6.34 trillion by 2035, a compound annual growth rate of 13.61%. The biologics consulting services segment specifically is projected to grow at roughly 15% CAGR through 2030, faster than the underlying biotech market, because complexity is outpacing internal capacity at most companies.
Three forces are accelerating that gap.
AI Is Reshaping Drug Discovery
AI-driven biotech is the headline story of 2025 and 2026. Isomorphic Labs (the DeepMind spinoff) raised a $600 million Series A in March 2025, and Xaira Therapeutics launched with over $1 billion in committed capital. Q1 2026 alone saw roughly $11 billion flow into AI drug discovery. The global AI-in-biotech market is forecast to grow from $3.12 billion in 2024 to $20.75 billion by 2035 according to Spherical Insights. Boards are increasingly asking consultants how to evaluate, partner with, or build AI capabilities — and that question now appears in nearly every strategy engagement.
Cell, Gene, and Advanced Therapies
Over 250 gene-editing clinical trials were active globally in early 2025. These programs require specialized regulatory, CMC, and commercial expertise that few companies have in-house, which is exactly the niche consultants are built to fill.
Capital Discipline
With biotech VC tighter than during the 2020–2021 boom, sponsors are running leaner teams and outsourcing more functions. Fractional CMOs, interim heads of regulatory, and project-based CMC support have all become standard rather than exotic.
Top Biotech Consulting Firms
The market is split between large strategy houses and specialist boutiques. Common names include:
- Strategy and management: McKinsey, BCG, Bain, Deloitte, EY, KPMG, Accenture
- Life sciences specialists: L.E.K. Consulting, ClearView Healthcare Partners, IQVIA, Syneos Health, ZS Associates, Charles River Associates, Trinity Life Sciences
- Regulatory and CMC boutiques: ProPharma Group, Alacrita, BioBoston Consulting, BBCR, and DES Pharma Consulting.
Large firms bring scale and cross-industry pattern recognition. Boutiques bring senior-only teams and deeper therapeutic specialization. Most well-run biotechs end up using both.
How to Choose a Biotech Consulting Partner
A few practical filters cut through the noise:
- Match the consultant to the milestone. A pre-IND gap assessment needs former regulators. A launch readiness review needs commercial operators. The skill sets do not transfer cleanly.
- Insist on senior delivery. Selling partners often pitch the engagement, but junior associates often deliver it. Confirm in writing who actually does the work.
- Check therapeutic depth. Oncology, rare disease, gene therapy, and neurology each have idiosyncratic regulatory and commercial dynamics. Generic playbooks fail.
- Demand documentation transfer. Good consultants leave you with templates, SOPs, and decision frameworks that strengthen your internal team after they leave.
The TLDR on Biotech Consulting in 2026 and Beyond:
Biotech consulting in 2026 is less about renting brainpower and more about renting execution capacity at the right level of seniority for the right milestone. As the underlying market climbs toward six trillion dollars and AI rewrites the discovery playbook, the companies that pair lean internal teams with sharply chosen external experts will keep moving while their competitors stall on avoidable problems.

Alex has 20+ years of experience in the CMC space, specializing in CDMO/CRO management, analytical development, technology transfer, quality and regulatory compliance for various drug modalities across multiple product stages.
Reach out to Alex on LinkedIn.



