Medical Safety/Pharmacovigilance

Because patient safety deserves expert focusMedical Safety and Pharmacovigilance
When medical safety expertise is critical and in-house capacity is limited, we serve as a trusted extension of your team—bringing the leadership, scale, and judgment needed to move programs forward with confidence.
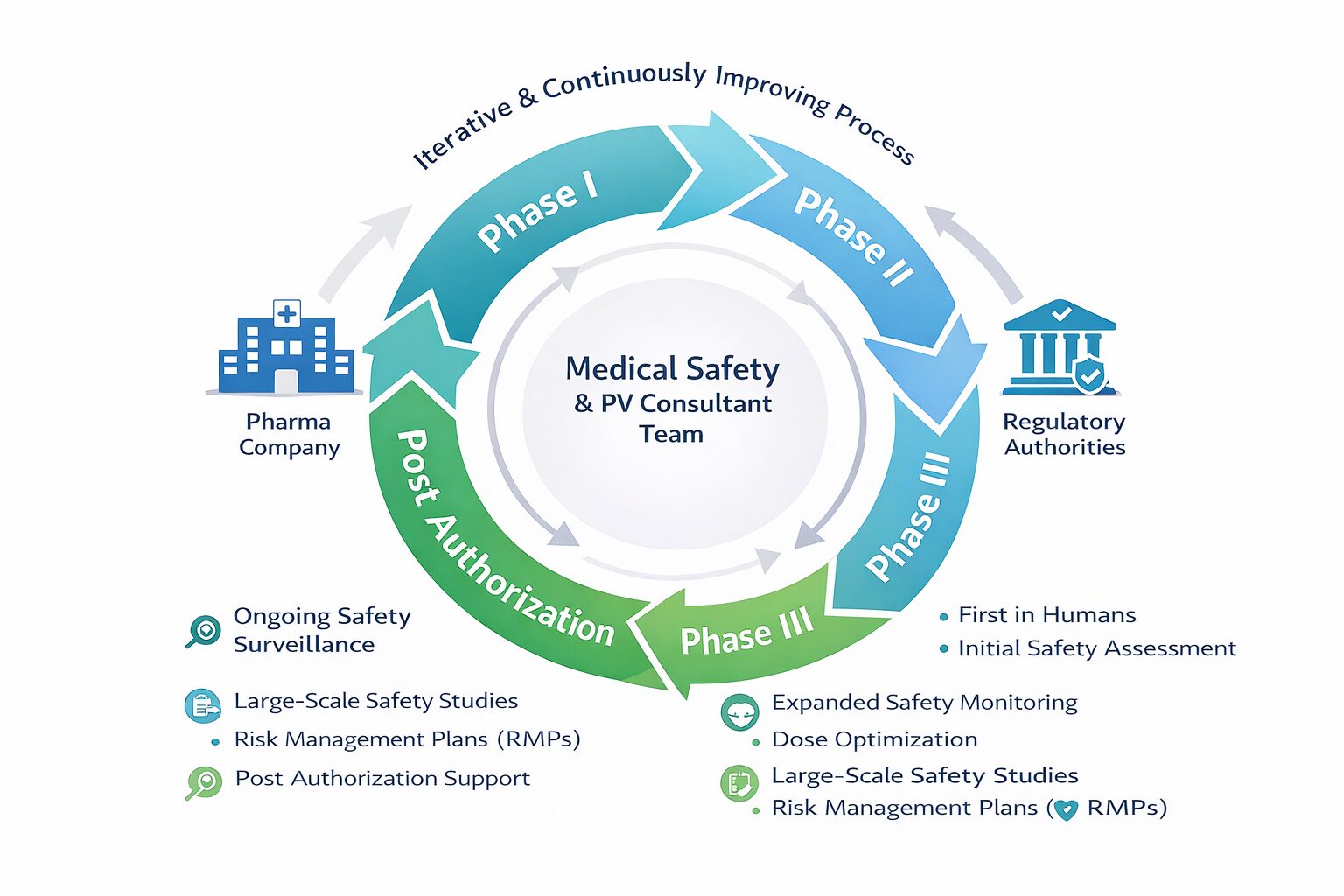
Phase 1/2:
Proactive signal detection, risk characterization, risk mitigation strategies, and communication..
- Establish medical safety strategies
- Regulatory safety strategies
- Safety portions of the target product profiles, study protocols, IBs, dose justifications
- Proactive safety monitoring and communication
- Drug monitoring committees
- Signal detection, risk mitigation strategies and implementation
- Aggregate reports
- Product profile for expanded indications
- Risk characterization, mitigation strategies, and implementation
- Marketing authorization application submissions
- QPPV
- Epidemiology
Phase 3/Post-Authorization
We drive Phase III and post-approval safety oversight that de-risks development, satisfies regulators, and protects patients.
- Continuous risk characterization, mitigation strategies, and implementation
- Independent drug monitoring committees
- RMPs, CCDSs, prescribing labels
Pharmacovigilance
We drive global, inspection-ready safety across Phase III and post-approval—combining compliant case management, blinded oversight, strong governance, and rapid regulatory response to de-risk development and enable your science to scale with growth.
- Pharmacovigilance Agreements
- Case processing, reporting, and oversight
- Quality and training
- KPIs
Processes for respective functions, implementation, and continuous oversight
Ensure compliance with local regulatory requirements at each phase
Regulatory authorities inquire responses
- Blinding processes for case processing and reporting, and oversight
- PSMF
- Expand capabilities for large-scale Phase 3 studies
Our teams combine strategic insight with hands-on execution to deliver medical safety solutions that support confident decision-making, regulatory success, and sustainable product growth.
Connect With Our Team TodayAdvance Your Drug Program
with Confidence
Start building reliable, high-quality drug development programs today.