
Disclaimer: This content is for educational purposes only and does not constitute legal or trade compliance advice. Consult qualified trade counsel before making tariff-related compliance decisions.
Where We Left Off: Recap of Key Supply Chain Themes
In our previous post, Navigating Critical Supply Chain Challenges in Pharmaceutical Manufacturing in 2026, we outlined the compounding pressures facing biotech and pharmaceutical developers: post-pandemic inventory normalization, escalating DSCSA serialization requirements, geopolitical API concentration risk, and intensifying cost pressures under the Inflation Reduction Act. We established that supply chain resilience is no longer a back-office function, it is a regulatory and competitive imperative.
Tariffs appeared in that discussion as a headline risk. In this post, we go deeper. With U.S. trade policy shifting dramatically (including a landmark Supreme Court ruling issued on February 20th 2026) pharma operators need a mechanism-level understanding of what is actually in force, what just changed, and what that means for their operations.
Tariffs 101 for Pharma Operators: Mechanisms, Not Headlines
Most supply chain teams track tariff news. The teams that navigate the ever changing tariff environment most effectively track tariff authorities and the legal mechanisms that determine who can impose what, under what conditions, and how it changes.
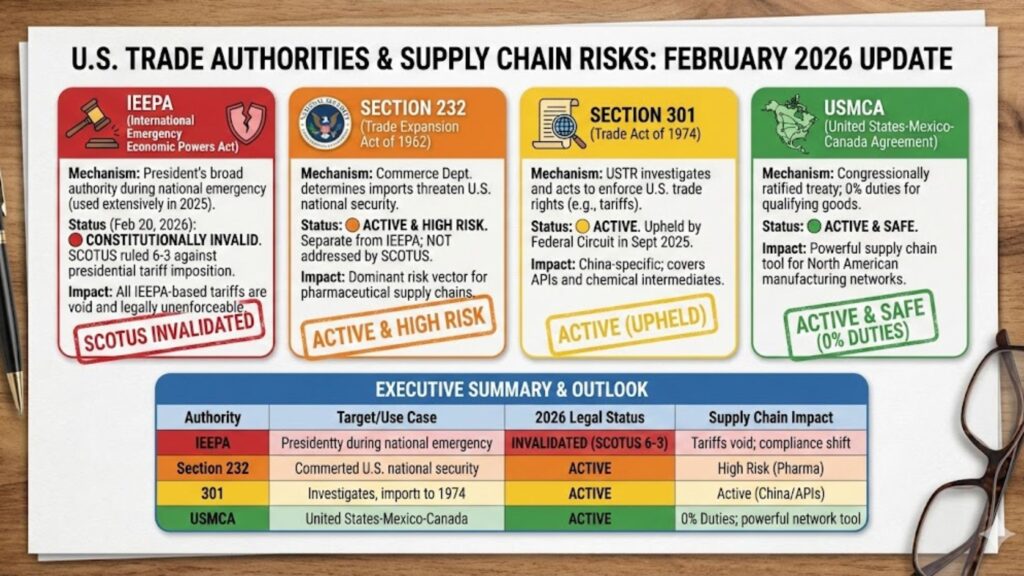
The U.S. currently has four primary legal levers for imposing duties relevant to pharmaceutical and biotech supply chains:
- IEEPA (International Emergency Economic Powers Act): Leveraged to provide the President broad authority to regulate trade during a declared national emergency. The Trump administration used IEEPA extensively in 2025 for country-specific “reciprocal” tariffs. As of February 20, 2026, the Supreme Court ruled 6–3 that IEEPA does not authorize presidential tariff imposition. All IEEPA-based tariffs are now constitutionally invalid.
- Section 232 (Trade Expansion Act of 1962): Authorizes tariffs where the Department of Commerce determines imports threaten U.S. national security. This authority is separate from IEEPA and was NOT addressed by today’s Supreme Court ruling. Section 232 remains the dominant risk vector for pharmaceutical supply chains.
- Section 301 (Trade Act of 1974): Grants the Office of the United States Trade Representative (USTR) a range of responsibilities and authorities to investigate and take action (e.g., impose a tariff) to enforce U.S. rights under trade agreements and respond to certain foreign trade practices. Primarily China-specific, covering APIs and chemical intermediates. Upheld by the Federal Circuit in September 2025.
- USMCA (United States-Mexico-Canada Agreement): A congressionally ratified trade treaty providing 0% duties for qualifying originating goods. USMCA compliance remains a powerful supply chain tool, particularly for North American manufacturing networks.
The implementing agency matters too. Customs and Border Protection (CBP) enforces at the border; the Federal Register publishes formal notices; the Harmonized Tariff Schedule of the United States (HTSUS), maintained by the U.S. International Trade Commission, is the definitive classification reference. For pharma operators, misclassification of an HTS code can mean the difference between a 0% and a 100% duty at the port of entry.
The 2025 Playbook: How IEEPA Was Used and Why It Mattered
Beginning in early 2025, the Trump administration deployed IEEPA to impose broad, country-specific tariffs, a use of the statute unprecedented in its scope. The administration cited border security and trade deficit concerns as the emergency predicates. Key 2025 IEEPA actions included:
- “Fentanyl” tariffs of on Canadian and Mexican imports, with pharmaceutical finished products and APIs initially included.
- “Reciprocal” tariffs on imports from most U.S. trading partners. Pharmaceuticals were carved out from these because of the ongoing Section 232 investigation.
- IEEPA escalation against China reaching >100%, subsequently reduced to 10% under a bilateral tariff agreement.
For pharma operators in 2025, the practical playbook centered on USMCA compliance. USMCA-qualifying goods were generally exempt from IEEPA reciprocal tariffs, making North American supply chains and rigorous country-of-origin documentation valuable assets. CBP simultaneously increased transshipment scrutiny to prevent Chinese or Indian APIs from being minimally processed in Mexico to gain USMCA certification without genuine substantial transformation.
As Jens Charles, Head of Transport and Trade Compliance at Swedish Orphan Biovitum, noted in a January 2026 DCAT Pulse podcast: three factors determine which tariff applies to any shipment: the HTS code (what the product is), the customs valuation (its declared value under specific customs rules, which differs from COGS), and the country of origin (where substantial transformation occurred). Getting any one of these wrong generates significant tariff exposure.
IEEPA and USMCA in 2025: What the Interplay Meant in Practice
The most consequential operational tension of 2025 was the relationship between IEEPA’s emergency tariff authority and USMCA’s treaty-based protections. Although IEEPA allowed the President to act instantly, USMCA is a congressionally ratified trade agreement and USMCA-compliant goods were generally shielded from IEEPA reciprocal tariffs in practice.
This created a significant compliance premium for companies with North American manufacturing networks, particularly those with Canadian or Mexican CDMOs, API suppliers, or fill-finish operations.
Today’s Supreme Court ruling fundamentally changed this paradigm. Because IEEPA tariffs are now constitutionally invalid, the USMCA-as-IEEPA-shield dynamic is no longer operative. However, USMCA compliance still matters for Section 301 (China) tariff management and will remain relevant if Section 232 pharmaceutical tariffs are formally proclaimed.
What’s Different Now: The Current Tariff Toolkit (February 20, 2026)
Today’s Supreme Court ruling is not the end of the tariff story for pharma. It is a plot twist that shifts the center of gravity entirely toward Section 232. Here is the operator-level summary of the current landscape:
2025 vs. Now: The Comparison Operators Need to Internalize
| Mechanism | 2025 Status | Feb 2026 Status |
| IEEPA (Emergency Tariffs) | Primary tool for reciprocal tariffs; applied to pharma finished products, APIs, and Canadian/Mexican imports (fentanyl orders). USMCA-compliant goods largely exempt. | STRUCK DOWN by U.S. Supreme Court (Feb 20, 2026). IEEPA does not authorize presidential tariff imposition. All IEEPA-based reciprocal tariffs are now invalid. Alternative tariff mechanisms may be employed. |
| Section 232 (National Security) | Investigation into pharma/APIs initiated in 2025. No formal pharma tariff proclamation issued by year-end. | ACTIVE and primary risk vector. Potential tariff on branded/patented pharmaceuticals announced with “Breaking Ground” construction exemptions. Formal Presidential Proclamation still pending. Section 232 survives SCOTUS ruling. |
| USMCA | Critical shield. USMCA-qualifying goods exempt from IEEPA fentanyl/reciprocal tariffs. CBP scrutiny increased for transshipment/country of origin. | Remains valid. No longer needed as a shield against IEEPA (now struck down), but still relevant for existing Section 301 (China) tariffs, and for any potential Section 232 impacts. |
| Section 301 (China) | Legacy tariffs on Chinese APIs and chemical intermediates. | ACTIVE. China tariff agreement reduces but does not eliminate Section 301 burden on Chinese-sourced APIs and starting materials. |
| MFN / TrumpRx Deals | Framework announced July–Sept 2025. Large Pharma companies have struck deals for 3-year Section 232 tariff holidays in exchange for U.S. investment commitments and MFN drug pricing. | 16 of 17 major companies signed as of Feb 2026. Product-level granularity critical: exemptions are company- AND product-specific. TrumpRx.gov portal active for tracking. |
The single most important takeaway: IEEPA-based reciprocal tariffs are gone as a legal matter, but Section 232, the dedicated national security track for pharmaceuticals, is unaffected and represents a potentially more permanent and structurally significant tariff mechanism.
Section 232 Today: What It Means (and Doesn’t) for Pharma Supply Chains
Section 232 is now the defining risk framework for pharmaceutical supply chains. Here is what operators need to understand with precision:
The Investigation and the potential 100% Tariff threat
The Department of Commerce initiated a Section 232 investigation into pharmaceutical imports and ingredients in 2025. There has been chatter of a 100% tariff threat on imported branded or patented pharmaceutical products. The stated basis was the ongoing Section 232 investigation and national security concerns about foreign pharmaceutical dependence.
The “Breaking Ground” Exemption
The primary operational exemption is the “Breaking Ground” rule: companies that have commenced construction in the United States are exempt from potential tariff threats. This has created a wave of announced U.S. manufacturing investments from major pharmaceutical companies.
For pre-commercial biotech companies, the Breaking Ground exemption is largely inaccessible. This makes the TrumpRx deal pathway and the MFN pricing framework the more operationally relevant exemption routes.
MFN Pricing: Clarifying a Term That Means Two Different Things
“Most Favored Nation” (MFN) appears in two distinct contexts in current pharma policy discussions, and conflating them creates serious confusion in supply chain and business planning:
- Trade MFN: The WTO principle requiring countries to apply the same tariff treatment to all trading partners (unless a preferential trade agreement exists). This is the baseline framework for standard duty rates at HTSUS — the “General” rate column applicable to WTO members.
- Drug Pricing MFN: The Trump administration’s policy requiring pharmaceutical companies receiving TrumpRx tariff exemptions to offer Medicaid programs pricing no higher than the lowest price offered in any other G7 country. This is a domestic pricing commitment, not a trade mechanism, though it is contractually tied to tariff relief.
Under the TrumpRx framework, large global pharmaceutical companies have signed agreements providing a three-year Section 232 tariff holiday in exchange for: joining the TrumpRx direct-purchase platform offering discounts to cash-paying patients; committing to drug pricing MFN with Medicaid; and making substantial U.S. capital investment. These deals are company-level and product-level.
For pre-clinical through Phase 2 companies: you are unlikely to qualify for or pursue a TrumpRx deal. Your tariff risk management must focus on HTS classification accuracy, country of origin compliance, and supply chain design — not executive deal-making.
How to Track Changes: A Practical Monitoring Framework for Pharma Operators
The single biggest operational failure we see is teams monitoring news cycles rather than authoritative sources. Here is a monitoring framework that builds a repeatable compliance intelligence function:
Tier 1: Primary Legal Sources (Check Weekly)
- Federal Register (federalregister.gov): The official record of all Section 232 proclamations, USTR notices, and CBP implementing guidance. Subscribe to alerts for NAICS codes and HTS chapters relevant to your products.
- HTSUS (hts.usitc.gov): Search your specific HTS codes against Chapter 99 (temporary/emergency tariff modifications) and the Pharmaceutical Appendix. This is the definitive document for what rate applies at the border today.
- CBP Trade Remedy Bulletins (cbp.gov): Official guidance on how duties are collected and enforced. Critical for country-of-origin disputes and transshipment risk.
Tier 2: Policy Tracking Resources (Check Bi-Weekly)
- TrumpRx.gov: Lists companies and products with active tariff exemptions under MFN deal frameworks. Product-level detail is critical for procurement decisions involving innovative therapeutics from major pharma counterparties.
- USTR.gov: Tracks Section 301 actions, country-specific deal terms, and modifications to reciprocal tariff frameworks.
- Congressional Research Service (CRS) Tariff Tracker (congress.gov): Comprehensive, frequently updated summary of all active tariff actions with legal citations. Excellent reference for briefing leadership and regulators.
Tier 3: Industry Intelligence (Monthly Review)
- DCAT Value Chain Insights (dcatvci.org): Industry-specific tariff analysis including pharma operator perspectives. The DCAT Pulse podcast is a high-signal resource (referenced in this post).
- BIO and PhRMA policy updates: Industry association filings and public comment submissions on Section 232 investigations are available publicly and often preview forthcoming regulatory positions.
- Major law firm Tariff Trackers: Firms including Skadden, Baker Botts, and Covington maintain updated trackers with Federal Register cross-references.
Our Final Thoughts: The Tariff Environment Rewards Operators Who Understand the Mechanisms
Today’s Supreme Court ruling eliminating IEEPA tariffs is a dramatic development, but it does not reduce pharmaceutical supply chain tariff risk. It concentrates and clarifies it. Section 232 is the operative authority, and it carries the weight of a national security determination. Companies that were building resilience against broad reciprocal tariffs will need to redirect that work toward the more targeted, sector-specific Section 232 framework.
For biotech and pharma companies advancing therapies from IND through NDA/BLA, the compounding risk is clear: tariff-driven cost increases on APIs, consumables, and manufacturing inputs arrive at the same time as the most capital-intensive stages of development.
Building tariff resilience into your supply chain is not a trade compliance exercise it is a strategic imperative.
DES Pharma Consulting: Your Partner for Tariff-Resilient, Regulator-Ready Pharmaceutical Supply Chains
At DES Pharma Consulting, we actively monitor the evolving U.S. tariff landscape and translate policy changes into practical CMC and supply chain strategy for our clients. Whether you are assessing your current API supplier exposure, planning a tech transfer with cross-border implications, or building your supply chain documentation package for an IND or NDA/BLA submission, we bring the regulatory depth and operational experience to help you act, not just react.
We understand tariffs not as a news story, but as a supply chain design variable that intersects with GMP compliance, regulatory strategy, and program economics. When the landscape shifts we are already positioned to advise.


