
The Critical Bridge in Drug Development
Technology transfer stands as one of the most pivotal—yet often underestimated—phases in pharmaceutical and drug development consulting. It’s the bridge connecting successful laboratory processes to scalable, compliant commercial manufacturing. Without a robust tech transfer strategy, even the most promising drug candidates risk delays, cost overruns, and regulatory setbacks.
What Exactly Is a Pharmaceutical Technology Transfer?
At its core, tech transfer involves systematically scaling laboratory-developed processes to production-scale manufacturing while maintaining product quality, safety, and regulatory compliance. This encompasses transferring formulation knowledge, manufacturing processes, analytical methods, and quality standards from development teams to manufacturing partners or contract manufacturers.
The stakes are high. According to the FDA, inadequate technology transfer is a common reason for manufacturing delays and product quality issues during scale-up.
3 Important Challenges in Tech Transfer
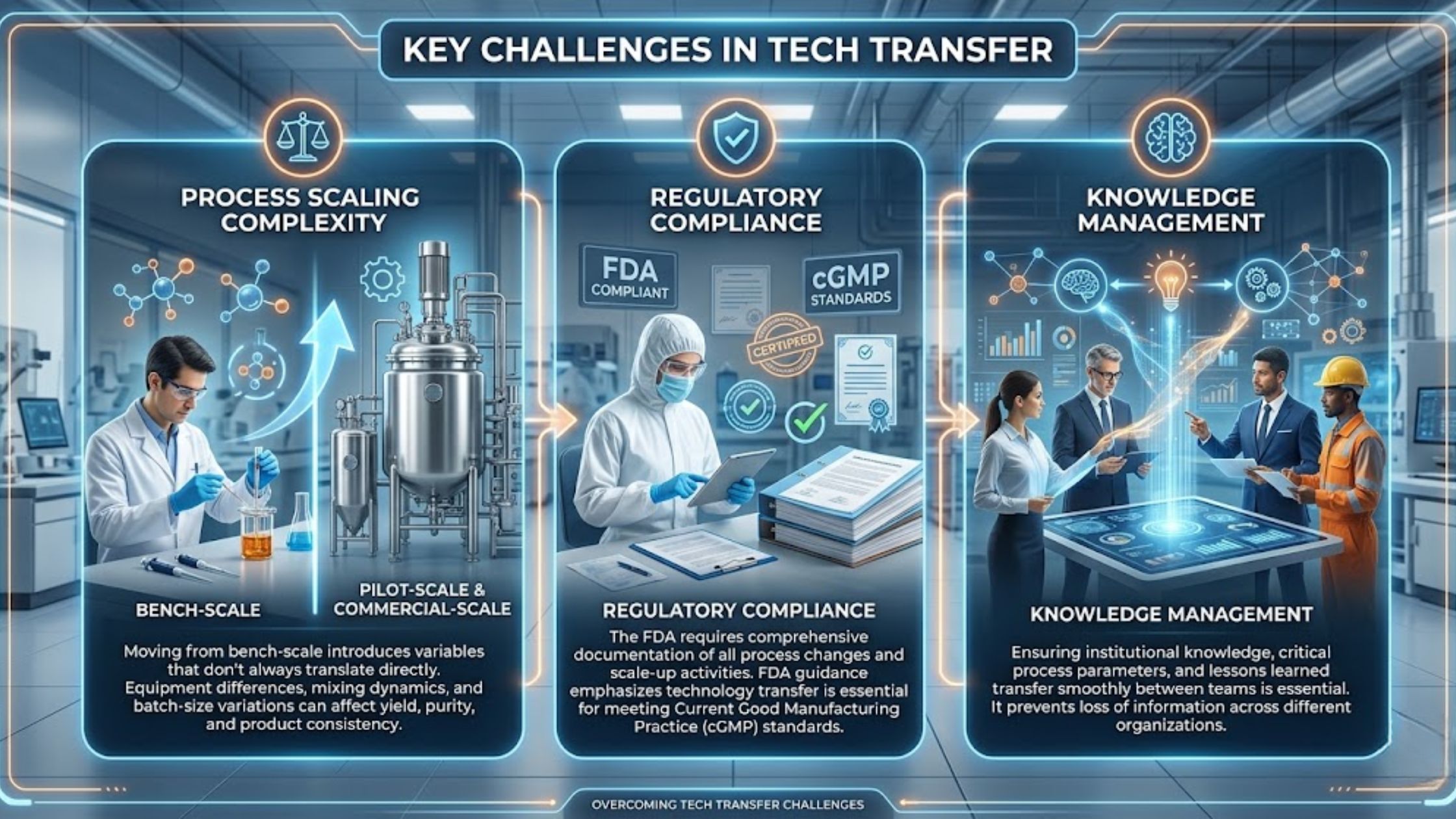
- Process Scaling Complexity Moving from bench-scale to pilot-scale and ultimately commercial-scale introduces variables that don’t always translate directly. Equipment differences, mixing dynamics, and batch-size variations can affect yield, purity, and product consistency.
- Regulatory Compliance The FDA requires comprehensive documentation of all process changes and scale-up activities. FDA guidance on manufacturing emphasizes the importance of well-executed technology transfer in meeting Current Good Manufacturing Practice (cGMP) standards.
- Knowledge Management Ensuring that institutional knowledge, critical process parameters, and lessons learned transfer smoothly between teams—often across different organizations—is essential for success.
Best Practices for Successful Technology Transfer
1. Plan Early Begin technology transfer planning during development, not as an afterthought. Define clear acceptance criteria and success metrics upfront.
2. Comprehensive Documentation Develop detailed batch records, process validation protocols, and manufacturing instructions that leave no room for interpretation.
3. Risk Management Identify critical process parameters and design robust manufacturing processes that are resilient to minor variations.
4. Cross-Functional Collaboration Engage development, manufacturing, quality, and regulatory teams early and throughout the process. Effective tech transfer requires coordinated, transparent communication.
5. Scale-Up Studies Conduct systematic scale-up studies to demonstrate that your process performs consistently at each scale. This data supports regulatory submissions and instills confidence in manufacturing capability.
Partnering for Success
Technology transfer doesn’t have to be navigated alone. Experienced consulting partners can help design efficient transfer strategies, conduct process validation, and ensure compliance with regulatory expectations. Working with specialists familiar with ICH Q7 guidelines and FDA manufacturing standards can significantly reduce risk and accelerate timelines.
The difference between a smooth, efficient tech transfer and a prolonged, costly one often comes down to preparation, expertise, and collaboration. Getting it right ensures your drug reaches patients faster and more reliably.
For authoritative guidance on pharmaceutical manufacturing and technology transfer, visit the FDA’s Drug Manufacturing Guidance and ICH Q7 Guidelines on good manufacturing practice for active pharmaceutical ingredients.

Alex has 20+ years of experience in the CMC space, specializing in CDMO/CRO management, analytical development, technology transfer, quality and regulatory compliance for various drug modalities across multiple product stages.
Reach out to Alex on LinkedIn.



